LADICIM collaborates with Valdecilla and IDIVAL to prevent structural failure of the heart and skeleton
Researchers from the University of Cantabria forge an alliance with clinical specialists to translate strict material fatigue methodologies to living tissue, transforming the treatment of valvular and bone pathologies
The laws of mechanics do not distinguish between the imposing structure of a viaduct and the intricate human anatomy. A femur withstands cyclic bending loads in a manner almost identical to a concrete pillar. The heart’s valves rely on tissue that tenses and relaxes millions of times, emulating the fatigue behavior of high-strength steel cabling. Based on this physical equivalence, researchers from the Laboratory of the Division of Materials Science and Engineering (LADICIM) at the University of Cantabria have consolidated a line of work alongside medical teams from the Marqués de Valdecilla University Hospital (HUMV) and the Marqués de Valdecilla Research Institute (IDIVAL). The goal is to apply the science of material fracture to the biological environment to decipher, and eventually predict, the collapse of human tissues.
This synergy, which began more than 15 years ago under a National Research Plan project to study cardiovascular behavior, has matured to forge an authentic ecosystem of multidisciplinary translational research. Today, cardiologists, traumatologists, and materials scientists share microscopes, traction machines, and artificial intelligence models in a joint search for answers that none of their disciplines could find in isolation.
Interestingly, the first obstacle to overcome in this collaboration was not technological, but communicative. The fields of materials engineering and clinical medicine employ drastically different nomenclatures and approach problem-solving from almost opposite philosophies. “At first, communication was very complicated due to a linguistic issue. Obviously, different specialties use different languages, and it always takes time to adapt,” explains Diego Ferreño, Professor at the University of Cantabria and LADICIM researcher. “The need to tear down that wall led us to organize joint summer courses in Santander and Burgos. There, away from the urgency of the laboratory or the operating room, the foundations of a common vocabulary were laid.”

Even beyond language, there was a profound discrepancy regarding the object of study itself. Once manufactured, metal or polymers begin an irreversible process of wear and tear. Biology, on the other hand, is alive. “Living organisms do not behave like inert objects,” emphasizes Professor Ferreño. “Natural selection has endowed us with repair mechanisms that rarely occur in inert nature. And that changes the rules of the game. Personally, I have learned a lot working with doctors.”
The physical convergence of both disciplines generated unusual scenes that are now part of the group’s history. Dr. José Manuel Revuelta, Professor Emeritus and Chair of Cardiovascular Surgery at HUMV, vividly recalls his first contact with the engineering environment: “I will never forget the morning I arrived at one of the LADICIM laboratories, full of train rails being tested in large machines… and I was carrying a jar containing a heart.”
Dynamics of mitral valve collapse
One of the pillars of this joint research has focused on unraveling the mechanics of the mitral valve. To understand its importance, it is essentially a kind of one-way safety gate located on the left side of the heart. Its function is to open to allow oxygen-rich blood to pass through and then immediately close tightly when the heart pumps forcefully, thus preventing blood flow from leaking back toward the lungs.
With an ever-increasing life expectancy, the aging of these tissues has become a significant challenge. Various diseases progressively deteriorate the leaflets (the “doors” of this gate) and, critically, the chordae tendineae—the tiny “cables” that anchor the valve to the papillary muscle, preventing it from turning inside out like an umbrella in a gale. When these chords elongate or rupture, blood flows backward, causing severe mitral regurgitation that drastically impairs the patient’s respiratory and physical capacity.
To understand the physics of these ruptures, LADICIM researchers, led by professors José Antonio Casado and Isidro Carrascal, decided to subject human chords—extracted during repair surgeries—to quasi-static tensile tests. It was not a trivial process. It required the design of 3D-printed millimeter-scale grips to hold the tissue without crushing it, and the constant immersion of the samples in a Hanks’ solution at 37 ºC to replicate the temperature and chemical composition of human blood.
The data obtained revealed a dramatic decline in structural capabilities due to calcification [1]. While a functional chord extracted from a healthy donor withstands stresses with a Young’s modulus of 233 MPa —a measure that defines the material’s stiffness; that is, how much force it can withstand when stretching or deforming—, moderately calcified chords plummeted to just 43 MPa. In practical terms, diseased tissue loses up to five times its natural stiffness (becoming more “brittle” and less elastic) and its tensile strength collapses from 46 MPa to a mere 10 MPa.
“Our contribution from the field of materials science and structural engineering has been fundamentally methodological,” Professor Ferreño points out. “Doctors are not unaware of the fact that tissues experience structural failures, but they lacked experimental methods designed to rigorously quantify and interpret the mechanical strength of a component.”
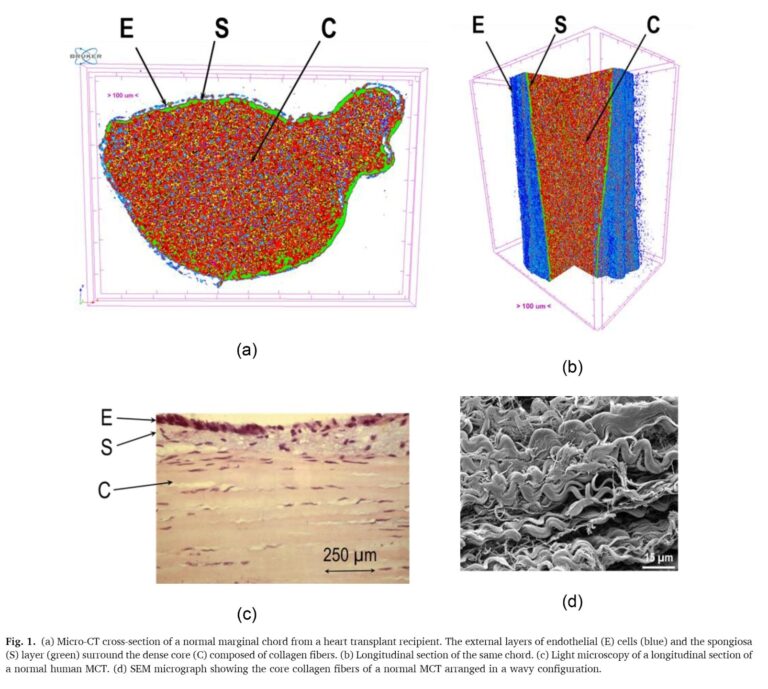
Thanks to the scanning electron microscope (SEM), LADICIM researchers visualized the disaster at a microstructural level. In healthy tissue, collagen fibers exhibit an elegant wavy pattern that allows them to absorb initial impacts before tensing. In calcified chords, hydroxyapatite deposits force these fibers to straighten permanently, grouping into rigid and brittle bundles that fracture abruptly under any hemodynamic overload.
Artificial intelligence to see the invisible
Far from settling for destructive testing, the team took a qualitative leap by incorporating Micro-Computed Tomography (micro-CT) combined with Machine Learning. The problem to be solved was of utmost urgency in the operating room: when repairing a mitral valve, the surgeon observes chords that appear completely healthy to the naked eye, but which may already harbor the onset of degenerative disease, compromising the long-term success of the operation.
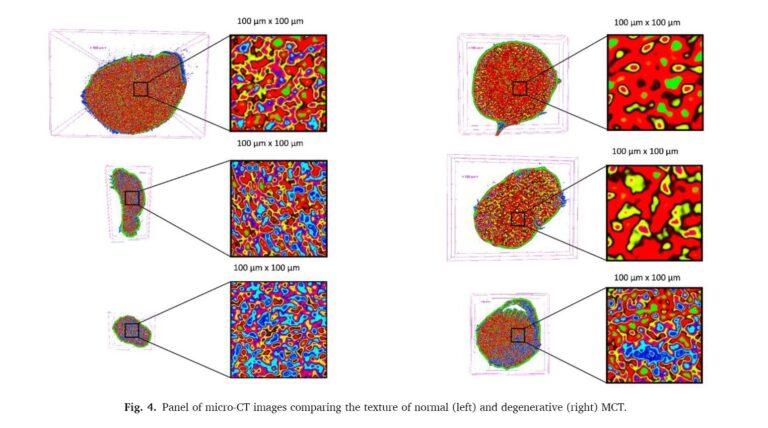
To “see” what the human eye ignores, researchers scanned 43 human marginal chords at a resolution of 2 micrometers (about 40 times thinner than a human hair). This allows for a virtual journey through the interior of the tissue without damaging it, obtaining thousands of digital slices of its intimate structure. Upon this immense data map, they applied the K-means algorithm, an artificial intelligence tool that automatically groups image points according to their density, identifying patterns that would be impossible to detect by sight alone.
To quantify this invisible damage, they turned to Shannon entropy. Entropy measures the level of “disorder” in a system. If healthy tissue is like a perfectly woven and structured fabric, diseased tissue resembles a tangled and chaotic ball of yarn. The results, published in 2022 in the journal Medical Engineering and Physics [2], conclusively demonstrated that diseased chords present significantly higher levels of entropy—that is, internal chaos. Thanks to this mathematical model, the system was able to differentiate between healthy and degenerate tissue with almost absolute statistical precision, detecting collagen disorder long before the surgeon can feel the weakness of the tissue.

In clinical practice, this discovery allows doctors to use entropy as a kind of predictive digital biopsy. By assigning a numerical value to tissue disorder, the surgeon no longer has to rely solely on intuition. If the analysis detects a high level of entropy, doctors know that the chord—even if it appears robust at that moment—is predestined to rupture in the short term. This enables the decision to replace it preventively during the operation, preventing the patient from suffering a relapse months later and having to undergo high-risk surgery once again.
Structural engineering applied to orthopedics
The analytical rigor of these trials led by LADICIM soon expanded into traumatology, working hand in hand with IDIVAL’s Bone and Mineral Metabolism Group. Led by Dr. José Antonio Riancho, Professor of Internal Medicine at HUMV, this team sought solutions for nonunion: fractures that, for various reasons, refuse to heal and leave the patient in a state of painful disability.
To find the most effective treatment, the researchers tested two drugs. “The ultimate goal was to enhance the fracture consolidation processes, especially in those patients who present delays in healing,” notes Dr. Riancho. To this end, the team compared a classic hormone (PTH) with strontium ranelate (SrR), working with a mouse animal model.
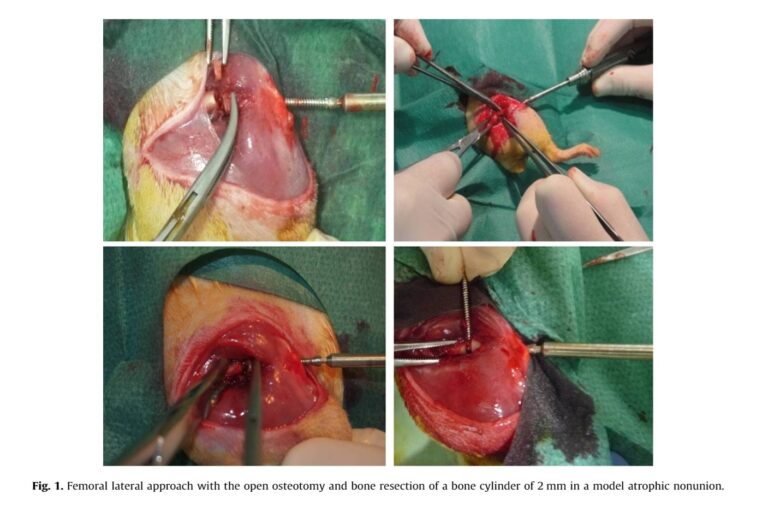
The definitive answer did not come from a simple X-ray, but from subjecting the bones to a true trial by fire in the engineers’ testing machine [3]. The results were conclusive: the group treated with strontium ranelate managed to forge a bone bridge so robust that it withstood a force of 61 Newtons, literally tripling the strength of the bones that received no treatment. Engineering demonstrated, with undeniable numbers, which medicine was building the strongest scaffold.
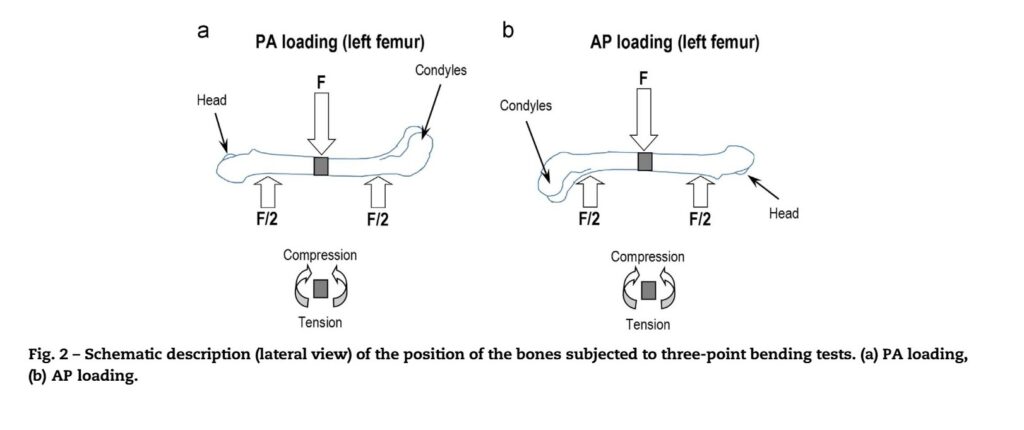
However, the most surprising turn occurred when materials engineers realized that medical science had been making a calculation error for decades. When testing the strength of a bone, laboratories worldwide typically placed it in the machine in any position, regardless of which side received the pressure. LADICIM experts suspected this was a mistake, as a bone is not a perfect cylinder, but an irregular and asymmetric structure.
To explain it simply: it is like trying to bend a plastic ruler. If you bend it along the flat side, it is easy; but if you try to bend it on its edge, it is much more difficult. The team demonstrated in a landmark study that, depending on whether the bone is supported by its front or back face, the results change drastically [4]. This geometric oversight had caused errors of up to 63% in thousands of previous studies worldwide. Thanks to this finding, LADICIM has established a new international standard: science now knows that to properly measure bone strength, orientation is everything.
The genome through a biomechanical lens
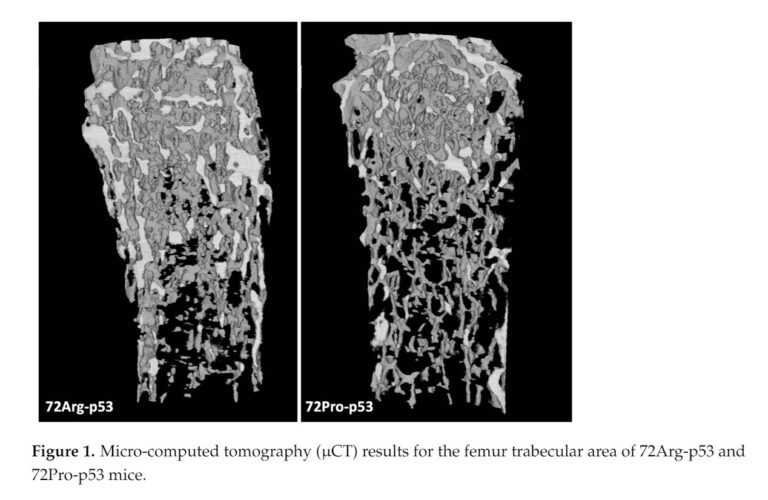
The synergy between engineers and doctors has recently reached a new level by entering the world of genetics. The challenge is to understand why, when faced with a similar fall, one person’s bone holds up while another’s breaks like glass. To this end, the consortium focused on a gene called TP53, known in medicine as “the guardian of the genome.” Its normal function is to ensure our cells are healthy; if it detects that a cell is damaged or has aged dangerously, it orders its removal to maintain tissue balance.
However, researchers discovered that a small variant in this gene (called Arg72Pro) drastically alters bone quality [5]. When analyzing this phenomenon with micro-CT, they observed that mice with this variant have a much weaker and more porous internal bone structure. The reason lies in the fact that the mutation weakens control mechanisms: the body produces fewer natural brakes against bone destruction and, furthermore, promotes the accumulation of senescent cells.
These senescent cells are biological units that have exhausted their capacity to divide but, instead of being eliminated, remain stagnant in the bone tissue. By staying installed in the bone without fulfilling their original function, they generate a kind of chemical interference, releasing substances that degrade and weaken the entire structure from within. “Being able to analyze how these genetic changes affect real bone strength has opened an immense door for us,” notes Dr. Riancho with satisfaction.
This line of research has even revealed unsuspected connections between our defenses and our skeleton. In a study with immunodeficient models [6], the team discovered that bone health depends on a constant dialogue with our immune system and our bacterial flora. In fact, they observed that, in certain cases, the absence of defenses prevented serious side effects in the jaw after using certain drugs. In other words: the solidity of our skeleton is not just a matter of calcium, but the result of a complex network of cells, genes, and bacteria.
Intraoperative technology: the near-term future
With methodologies consolidated in the static environment of the laboratory, researchers are already looking toward the horizon of real clinical practice. The imperative challenge is for engineering to step into the operating room to guide the surgeon’s hand in real time.
“In the near future, this modern technology will provide a tool that allows the surgeon to confidently diagnose which chordae tendineae are normal and which are affected by the degenerative process, even if they appear normal to the naked eye,” anticipates Dr. Revuelta.
The first step is already underway through a cross-disciplinary alliance with the Photonic Engineering Group of the University of Cantabria, led by professors José Miguel López Higuera and Olga Conde Portilla. Using Optical Coherence Tomography (OCT), they plan to develop an intraoperative sensor the size of a scalpel. The device, supported by the Shannon entropy algorithms previously calibrated by LADICIM, will evaluate collagen birefringence patterns in situ. A simple light scan will be enough for the software to dictate whether the mitral tissue will withstand cardiac pressure or if it must inevitably be removed.
Similarly, traumatology pursues its own algorithmic oracles. “The application of machine learning methods would be interesting for a better prediction of bone strength, both in vivo, based on radiological studies and other techniques, and in ex vivo samples,” says Riancho.

The metamorphosis experienced by LADICIM over the last two decades perfectly exemplifies the real value of transversal research. A department historically focused on homologating railway tracks, certifying the fatigue of large-scale steels, and validating construction materials has successfully channeled its powerful mathematical and structural heritage into the complex puzzle of the human body.
The shared journey of the University of Cantabria Laboratory and the clinical research facilities of the Marqués de Valdecilla Hospital yields a vital lesson: great technological leaps are born in the margins and frictions between seemingly distant sciences. Treating mitral degeneration or osteoporosis with the same analytical coldness required by the construction of a suspension dam does not detract from the humanity of medicine; on the contrary, it provides specialists with incomparably more precise tools.
In the short term, this torrent of empirical data promises tailored cardiac interventions, much more robust traumatological recoveries, and an undeniable improvement in the quality of life for thousands of patients who, without knowing it, benefit from the convergence of engineering and molecular biology.
Scientific References
- [1] Casado, J. A., Diego, S., Ferreño, D., Ruiz, E., Carrascal, I., Méndez, D., Revuelta, J. M., Pontón, A., Icardo, J. M., & Gutiérrez-Solana, F. (2012). Determination of the mechanical properties of normal and calcified human mitral chordae tendineae. Journal of the Mechanical Behavior of Biomedical Materials, 13, 1-13.
- [2] Ferreño, D., Revuelta, J. M., Sainz-Aja, J. A., Wert-Carvajal, C., Casado, J. A., Diego, S., Carrascal, I. A., Silva, J., & Gutiérrez-Solana, F. (2022). Shannon entropy as a reliable score to diagnose human fibroelastic degenerative mitral chords: A micro-ct ex-vivo study. Medical Engineering and Physics, 110, 103919.
- [3] Pérez Núñez, M. I., Ferreño Blanco, D., Alfonso Fernández, A., Casado de Prado, J. A., Sánchez Crespo, M., De la Red Gallego, M., … & Riancho Moral, J. A. (2015). Comparative study of the effect of PTH (1-84) and strontium ranelate in an experimental model of atrophic nonunion. Injury, 46, 2359-2367.
- [4] Ferreño, D., Sainz-Aja, J. A., Carrascal, I. A., Diego, S., Ruiz, E., Casado, J. A., Riancho, J. A., Sañudo, C., & Gutiérrez-Solana, F. (2017). Orientation of whole bone samples of small rodents matters during bending tests. Journal of the Mechanical Behavior of Biomedical Materials, 65, 200-212.
- [5] Usategui-Martín, R., Galindo-Cabello, N., Pastor-Idoate, S., Fernández-Gómez, J. M., del Real, Á., Ferreño, D., … & Pérez-Castrillón, J. L. (2024). A Missense Variant in TP53 Could Be a Genetic Biomarker Associated with Bone Tissue Alterations. International Journal of Molecular Sciences, 25(3), 1395.
- [6] del Real, Á., López-Delgado, L., Sañudo, C., García-Montesinos, B., Wert-Carvajal, C., Laguna, E., García-Ibarbia, C., Saiz-Aja, J. A., Ferreño, D., Casado, J. A., Menéndez, G., Pérez-Núñez, M. I., & Riancho, J. A. (2024). Limitations of immunodeficient mice as models for osteoporosis studies. Revista de Osteoporosis y Metabolismo Mineral (ROMM).
Más Noticias

LADICIM and Viuda de Sainz strengthen their collaboration for the development of recycled concrete
The Laboratory and the construction company are working together to consolidate the use of recycled aggregates in civil engineering, in line with European regulatory requirements

LADICIM is participating in the development of a new generation of floating concrete platforms for offshore wind energy
The ISOBARA project, in collaboration with SEAPLACE and IHCantabria, designs industrializable solutions to reduce costs in floating wind farms The energy transition toward a low-carbon

LADICIM Participates in the Development of Metropolitan Transport in India
The Laboratory of the Division of Materials Science and Engineering has renewed its collaboration agreement with Patil Rail, one of India’s leading companies specializing in
